HAE (hereditary angiodysplasia)Icatibant Acetate
Icatibant (Firazyr) is a synthetic peptidomimetic drug consisting of ten amino acids, and acts as an effective and specific antagonist of bradykinin B2 receptors. It has been approved in the EU for use in hereditary angioedema, and is under investigation for a number of other conditions in which bradykinin is thought to play a significant role. Icatibant currently has orphan drug status in the United States and FDA approved on August 25, 2011.
Icatibant Acetate is a selective competitive antagonist for the bradykinin B2 receptor. It is indicated for the treatment of acute attacks of hereditary angioedema (HAE). HAE is a rare disorder resulting in periodic attacks of acute edema. It is characterized by a deficiency of functional C1 esterase inhibitor that leads to the overproduction of bradykinin, which triggers vasodilation, vascular permeability, and edema. Icatibant Acetate inhibits bradykinin from binding to the B2 receptor and thereby treats the clinical symptoms of an acute, episodic attack of HAE.
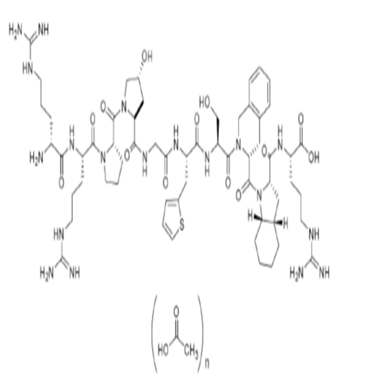
Icatibant Acetate Structural Formula:
Diseases and fields of Icatibant Acetate
| Product | Status | Indication of dosage form | Market |
|---|---|---|---|
| Icatibant Acetate | Commercial | HAE (hereditary angiodysplasia) | Overseas |
Specifications
| Synthesis mode | chemic synthesis,solid phase synthesis |
|---|---|
| Categories | APIsPeptides; Biopharmaceuticals |
| Sales markets | Western Europe; Asia; North America; Central/South America |
| Supplied from | China |
| Selling Points | International Approvals/Standards |
Show more specificationsConsultation details
Other Polypeptide APIs Products
peptide synthesis companies
| Polypeptide APIs Products |
| US-DMF LIST |
| Beauty peptides |
| Chinese cGMP APIs |
| Mexico Registered APIs |
| Research Peptide APIs for Regulatory Market |
| Polypeptide Preparation |
| Kaijie bio medicine Peptide APIs |
How many companies are there in peptide api manufacturer in china? The peptide api market is very promising, and the world is encouraging the development of peptide business. There is a peptide api list on the website Biofda.com, which contains various specifications of peptide APIs for customers to choose from. Shengnuo Technology is a peptide api manufacturer located in Chengdu, a city in southwest China. Not only peptide APIs, but also carnosine custom suppliers and cosmetic peptide suppliers
There are many peptide apis manufacture in China, but they are all small-scale companies. The China peptide company such as Sinotech is a leading company in China and has a very high position.
As a Chinese peptide company, Sinotech has been working silently, hoping to become a top peptide company in the world. There are many countries producing peptides in the world, such as bulk drug substance in India, gmp custom peptide in uk, and peptide production in usa. So what is polypeptide? What kind of peptide synthesis supplier should you choose? Follow our website: www.biofda.com, here will tell you the answer.